Introduction and history of covalent bonds
The idea of covalent bonding was first introduced by the American chemist Gilbert Lewis in 1916. This was only shortly after scientists had begun finding out about the particles inside atoms and while they were still trying to piece together a clear model of atomic structure. It was less than 20 years since Thomson discovered the electron, only around 5 years from Rutherford’s discovery of the nucleus, and just a year after Moseley discovered the proton.
Lewis’ model of covalent bonds
The understanding of electrons that existed when Lewis presented his ideas pre-dates many features we are now familiar with, such as details of electron orbitals and the Pauli Exclusion Principle. Parts of his model might seem odd to us now, but the idea and method for understanding bonding still works well. For example, Lewis pictured atoms with an outer shell of electrons arranged at the corners of a cube. Different elements had different numbers of electrons in their outer shell and so different numbers of empty corners. Bonds could form between atoms, joining them together into groups called “molecules”.
Based on a great number of observations made over many years, it was apparent that particular elements often formed the same number of bonds when joining with others to become molecules. Lewis proposed that he could explain this by imagining atoms bonding together by sharing pairs of electrons, one from each atom, until all the corners of their cubes were filled with electrons. Elements with more empty corners would therefore share a greater number of electron pairs and (therefore form different numbers of bonds) in order to complete their cubes. This would usually result in atoms having 8 electrons, one in each corner of a cube.
Here is the way we could understand a chlorine molecule, Cl2, forming by each atom contributing one electron to a covalent bond and both atoms have completed sets of 8 electrons.
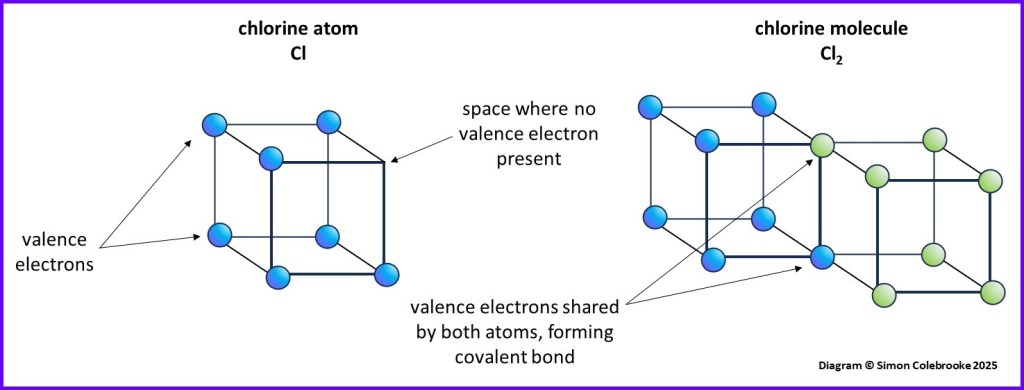
Lewis also introduced a style of diagram to summarise how many electrons each atom had and shared with others. A series of dots represented the electrons in the outer shells of the atoms in a molecule, drawn between the element symbols of bonded atoms – which signified the nucleus and inner electrons.
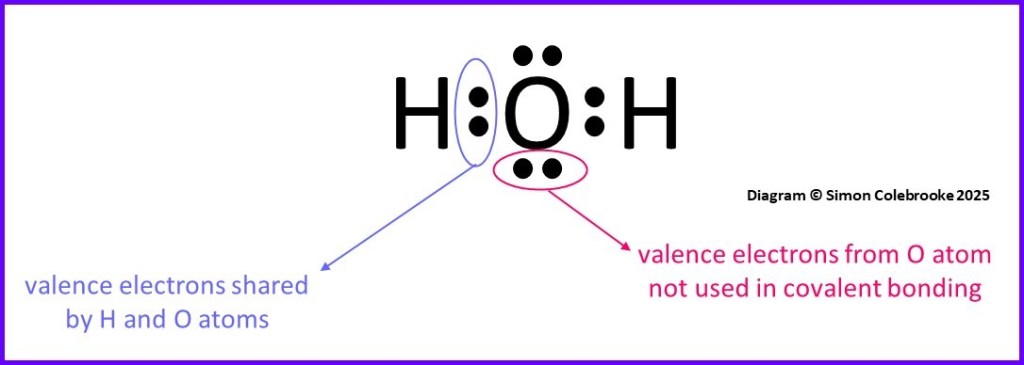
We keep many of these ideas today; the number of electrons that elements have in their outer shell is the same, we still treat covalent bonds as shared pairs of electrons and very often, atoms usually have 8 electrons represented around them when we draw “dot-cross” diagrams. However, our understanding of atomic structure and electron orbitals means we don’t view atoms like cubes any longer.
Irving Langmuir developed the ideas of Lewis in 1919 and first introduced the term “covalence” to mean the number of pairs of electrons an atom shares with neighbouring atoms.
The “octet rule”
The idea that atoms form covalent bonds so that they obtain a share of 8 valence electrons is often referred to as the “octet rule”. Hydrogen because it is only using the first shell of electrons forms compounds where it has a share of two electrons rather than eight. It was an extremely useful development because it successfully accounted for the proportions of which many elements combined together to make molecules. Molecules from so many different areas of chemistry appear to follow this rule and it is well worth using it in many cases, during A-level. Using the octet rule will very often result in us working out the formula and structure of a molecule correctly. The next section will illustrate in more detail how the octet rule can help determine the number of bonds present in a range of different molecules.
However, in the 100 or so years since Lewis published his first article many new compounds have been discovered, that push the boundaries of how elements form covalent bonds, so in later pages we will look at electron rich and electron deficient molecules and need to develop a more detailed model of bonding.
Electrostatic forces in covalent bonds
As described above the word covalent means sharing electrons (“co” meaning joint / shared and “valent” meaning outer shell electrons). But why should atoms share electrons ?
Change in electrostatic forces with distance
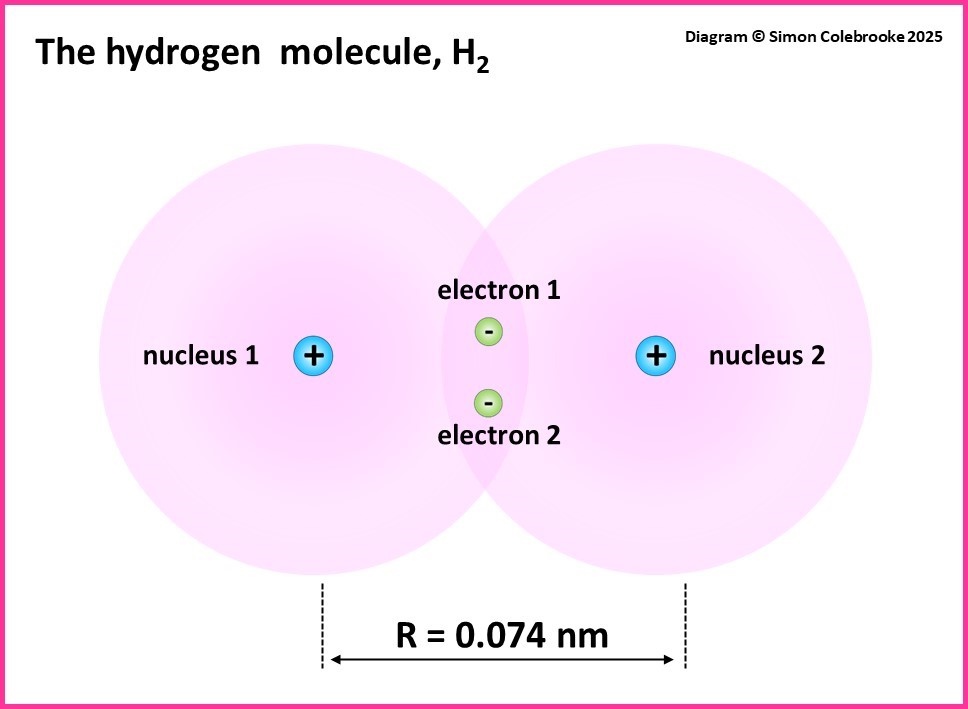
We can get some insight into this by thinking about the electrostatic forces that will arise when two atoms start to get closer together. The simplest atoms are hydrogen atoms with mass of 1 and we can imagine a “system” of two hydrogen atoms separated by a distance “R” between their nuclei.

First imagine that the atoms are extremely far apart and R is therefore very large. Any electrostatic forces between the charged particles that make up these two atoms (a proton and electron in each) will be negligibly small. This is because the size of the force between two charged particles is inversely proportional to the square of the distance between the charged particles. We can say that the combined potential energy of the atoms with this separation is zero.
If the two atoms start to get closer together (and the distance decreases) then the electrostatic forces become larger. Both attractions and repulsions will occur:
Repulsive forces between particles with the same charge, for example the electrons from the two separate atoms. These will increase the potential energy of the system, effectively making it less stable. There will also be repulsions between the positive nuclei of the two atoms, most significant when the atoms get very close.
Attractive forces between particles with opposite charges, in the case electrons and nuclei. The electrons will be attracted to both nuclei at once.These attractions will reduce the potential energy of the two-atom system and increase the stability.
These forces are shown in the diagram below:
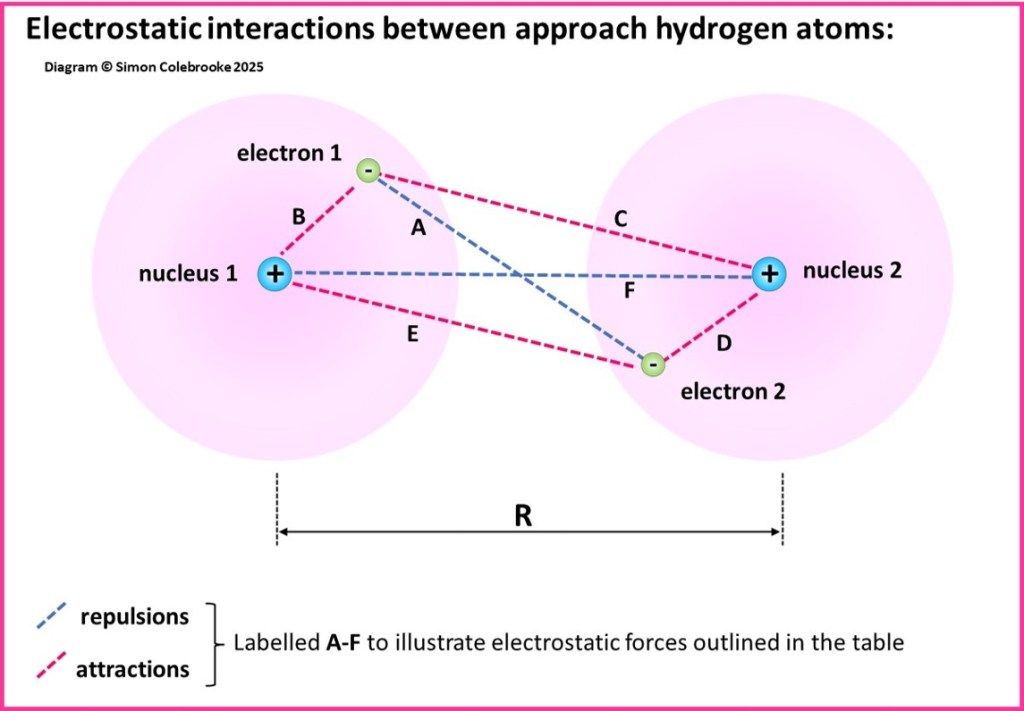
Even for an atom as simple as H2, we can think of six possible electrostatic forces between the various charged particles that are present:
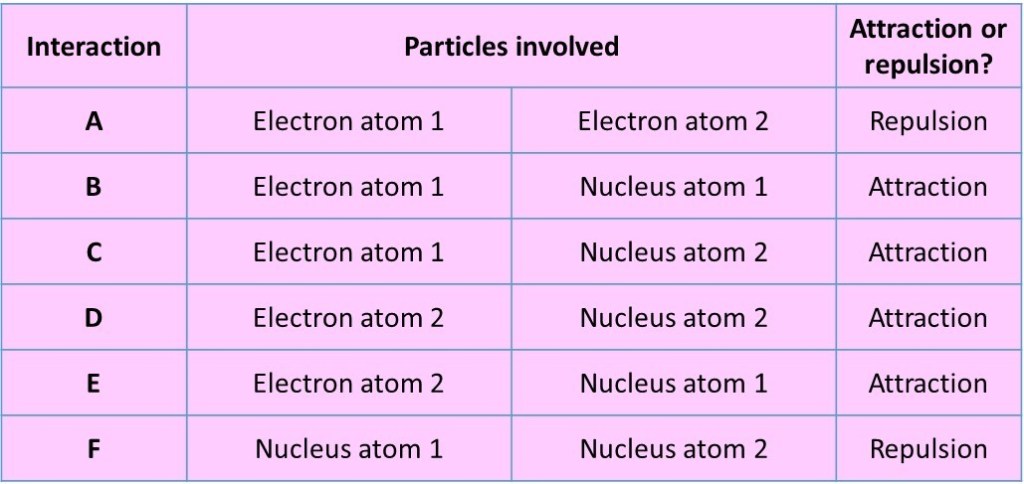
As the atoms start to approach each other, the attractions are greater than the repulsions for a while so that, overall, the two atoms move to lower potential energy than when they were much further apart. This means that the system becomes more stable. The attractive forces are maximised if the electrons are primarily located between the two approaching nuclei.
However, the rate at which the attractions and repulsions change is different. In particular, when the atoms are extremely close, the repulsive forces increase very rapidly as R decreases further, due to increasing repulsion between the two positive nuclei. The potential energy of the system now increases very quickly and the energy becomes much higher than it was for the two separated atoms.
Potential energy minimum for covalent bond
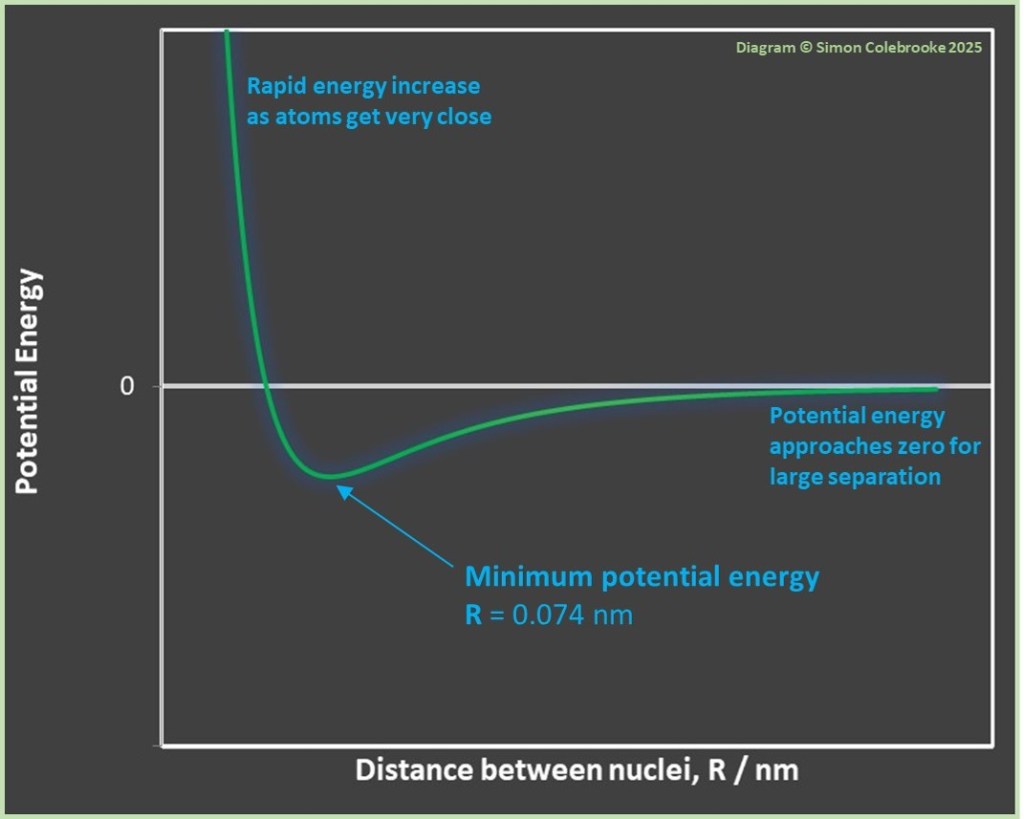
Overall – adding the effect of repulsive and attractive forces – a minimum potential energy is obtained at a specific separation for distance R. For the H2 molecule this minimum occurs with the nuclei separated by 0.074 nm, which is a significantly shorter separation that would be obtained by adding the radii of two touching (but not bonded) hydrogen atoms.
The graph below shows how the potential energy of the system varies as the separation of the two atoms, R, changes. The position of the minimum is indicated. The minimum means that the atoms are more stable when at this separation than when they are either further apart or closer together. The position of lowest potential energy represents the position in which the system is most stable and the strongest bond is formed*. The fact that the potential energy gets lower as atoms approach one another means it is favourable for the atoms to bond.
*The separation R is actually not rigidly fixed; bonds vibrate so that the atoms continually oscillate to get slightly closer or further apart – the minimum shows that average separation at the most stable state.
As the system has lower energy when the atoms are bonded together, the formation of the covalent bond releases energy. Alternatively, energy will have to be supplied in order to increase the separation of the atoms and break the bond.
All other molecules are more complicated than H2, as they consist of more charged particles and so involve many more attractive and repulsive forces. However, it is still useful to imagine an equivalent situation, where a minimum potential energy occurs at a certain separation and attractive forces between electrons and nuclei dominate.
Comparison to Lewis’ model of covalent bonds
Lewis described covalent bonds as involving atoms sharing electron pairs. We can now picture this sharing by imaging the two electrons being attracted to nuclei from two different atoms – they are “shared” by interacting with both nuclei simultaneously. It makes sense to imagine the electrons are mostly located between the two nuclei as this would maximise the electrostatic attractions, which is kind of indicated in the dot-cross diagrams.
Limitations
The picture above is a simplified one, useful for giving us an insight into how an attraction between two atoms could arise. It is simplified even for a very basic molecule like H2. For example, it doesn’t include changes in the kinetic energy contribution to the system, particularly for the electrons. None-the-less, it does give us a way of understanding some of the factors involved in forming a covalent bond and also why bonds have a certain length (important in many areas of chemistry).
A later section will look at an alternative way of understanding covalent bonds, based on a more quantum mechanical approach and orbital overlap. This method will account for some additional features for example:
Why do covalent bonds usually involve pairs of electrons, rather than some other number?
And:
Why does the potential energy increase so rapidly for small inter-nuclear separations, R?
Conclusion – covalent bonds still being researched
This simple model of covalent bonding will get us started in understanding how and why molecules form and give us a way of starting to explain the properties of some materials. So treat this section as a way of gaining some insight into covalent bonds, rather than explaining the properties of molecules fully.
Believe it or not, the exact nature of covalent bonding is still undergoing research, 110 years after Lewis’ ideas were published! As recently as the 2020s, articles discussing the interactions and factors involved have been produced. In addition, chemists are always discovering new and unusual molecules with exotic covalent bonds – like bonds with 1 shared electron rather than 2, bonds spread over three atoms (in B2H6) and delocalised electrons in covalent molecules…. so an active area research, for which our understanding will develop in the future.
“Copyright Simon Colebrooke 20th November 2025″
In writing this article I referred to the recent publication “The basics of covalent bonding in terms of energy and dynamics” by Nordholm and Bacskay, published in the journal “Molecules” in 2020, 25, 11, 2667.
The original Lewis article was also useful; “The atom and the molecule”, Gilbert N. Lewis, Journal of the American Chemical Society, 1916, 38, (4), 762-785.
Click the icons below to either return to the homepage, or try a set of questions on this topic (choose the Q icon) or return to the notes menu (N icon).
 chemistryexplained.uk
chemistryexplained.uk

