Previous sections have described the method for preparing a standard solution (solution with known concentration) and the types of errors that can be avoided by following the procedure carefully. This section will outline how you can calculate the concentration of a standard solution.
Before giving specific examples, it is worth pointing out that some solids can be reliably used to produce a standard solution – for example potassium carbonate – as they do not absorb or lose moisture from the air. These are called “primary standards“. Other solids may absorb / lose moisture or interact with other substances in the environment and that will affect how well the mass measurements made in the procedure actually reflect the amount of solute used. So it is important to check whether your solute can be considered a primary standard – if not, then additional procedures might be needed to determine the concentration. For the purposes of this article, I will assume that all solids are in a condition where they can be directly use to make a standard solution.
Concentration units
Concentration means “amount of solute per volume of solution”. The most commonly used used units are mol dm-3 (moles of solute per decimetre cubed of solution) and g dm-3 (grams per decimetre cubed of solution). Although other sets of units are used as well (simply change the quantity that the amount or volume are measured in), they are all essentially the same thing – an amount of solute divided by the total volume into which it is dissolved.
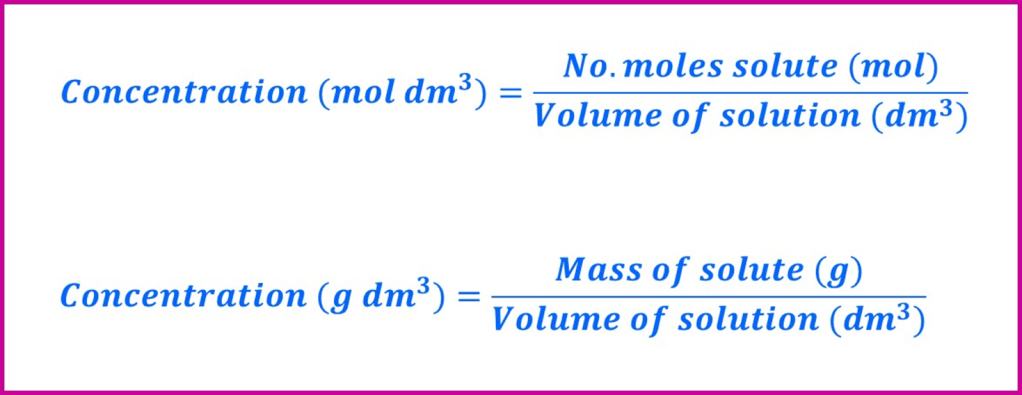
Stages of the calculation
When you make a standard solution you will always have a concentration in mind that you wish to achieve. When the correct procedure is followed the final concentration of the solution ought to be very close to this value, but will almost certainly be a little different, due to the method required to ensure you know exactly how much solute was used.
Hence there are really two separate calculations to do:
i) An initial calculation based on the desired concentration you want your final solution to have. This must be done before the solution is made so that you know roughly what mass of solute needed.
ii) A final calculation of the actual solution concentration the solution has, once the preparation is complete.
A few examples will illustrate these points.
Example 1: Preparation of 100 cm3 of 0.200 mol dm-3 sodium chloride (NaCl) solution
i) We need an initial calculation to determine the mass of sodium chloride to dissolve. This is obtained by some relatively simple calculations, based on equations linking moles, volume and concentration and also moles, mass and molar mass. First, we calculate the number of moles of NaCl needed, based on the concentration and volume of solution required. We then use that result to calculate the corresponding mass of NaCl. All the steps are shown in the box below:

Given the method described in the previous sections it is not realistic to measure a mass of 1.170g and know for certain that it has all been transferred into the solution. Apart from anything else, it would take too long to add the correct amount of NaCl to the weighing boat at the third decimal place – we’d constantly be adding too much, then trying to remove a bit, only to take off too much and have to add more again! Instead, masses are measured by difference, with approximately the correct mass added to a weighing boat and, once the solid has been tipped into a beaker, the mass of the weighing boat recorded again with the difference between the two measurements equalling the mass added to the beaker. This accounts for miniscule amounts left on the weighing boat…. And it’s much faster!
However, we use the calculated mass of 0.170g as a guideline for how much to add, in the following way:
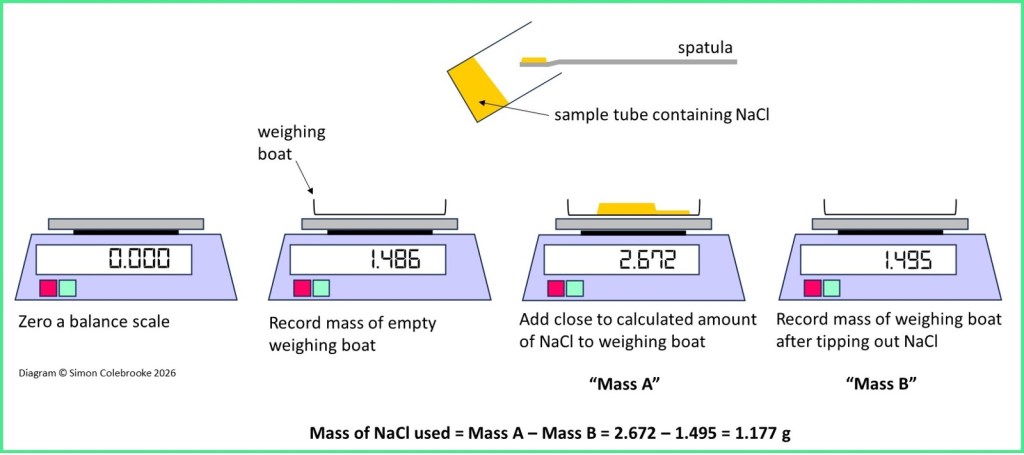
- Zero the balance.
- Add an empty weighing boat and observe the mass. In my example 1.486 g.
- Without zeroing the scale again, start adding sodium chloride to the weighing boat, observing the balance which now reads the combined mass of the weighing boat and any added NaCl. In this example, if the combined mass reached 2.656 g we would have added exactly 1.170 g (since 1.486+1.170 = 2.656 g). However, in reality we only need to aim for something close to that, for example between around 2.620 – 2.680 g. It doesn’t particularly matter what the total mass is so long as we record exactly what it is. There is no point in us spending time getting the third decimal place to be equal to one particular value; we won’t be able to transfer all of that solid into a beaker anyway and it is impossible to guess how much will be left on the weighing boat. So, being a bit higher or lower than 2.656 g doesn’t matter, we just need to get to a position where we know exactly how much NaCl is tipped out into the beaker.
- Once we are in the required range record the combined mass of the weighing boat and NaCl. This is mass A. In the example, the combined mass is 2.672 g.
- Tip out the NaCl into a beaker for the next stage of the method.
- Re-weigh the weighing boat, which will almost certainly be a higher mass than the original empty boat, due to small pieces of NaCl that are left behind. This is mass B, which in the example is1.495 g and clearly higher than the original clean weighing boat.
- Find the actual mass of NaCl used, by finding the difference between mass A and mass B. In the example this is 1.177 g, so a little bit higher than the calculated mass for 0.200 mol dm-3, but perfectly suitable to give a solution with very similar concentration.
The process is summarised by the diagram below showing the relevant balance displays:
ii) Having measured exactly how much NaCl is used in the preparation of this solution, follow the steps outlined in the procedure described previously to obtain 100 cm3 of solution in a volumetric flask. It is now possible to calculate the concentration of the solution actually prepared, as shown in the box below:

The sodium chloride example used here illustrates how weighing by difference allows the concentration of the standard solution to be precisely known and avoids a very painstaking weighing process to add an exact amount to the weighing boat.
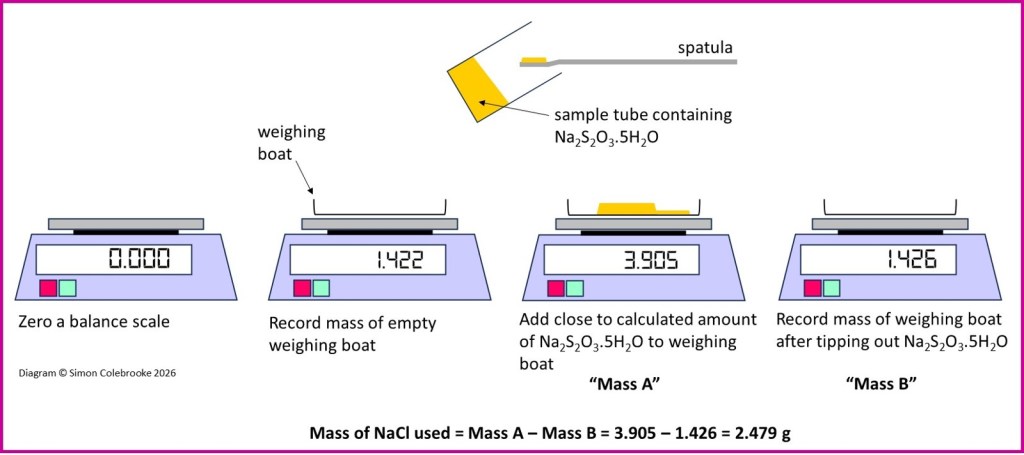
Example 2: Preparation of 250 cm3 of 0.040 mol dm-3 sodium thiosulphate from the hydrated salt Na2S2O3.5H2O.
The same two calculations are required for this solution; i) the initial calculation of the desired mass and then ii) a calculation after the preparation to determine the actual concentration obtained. These steps are shown in the two boxes below, along with typical balance scale values.
Here is the initial calculation:

However it is important to think about the formula of the hydrated salt before starting the calculation. The crystal lattice of a hydrated salt includes some water molecules. This is different to the crystals being “wet” on the surface, the water is actually incorporated inside the crystal amongst the sodium and thiosulphate ions. So when we determine the mass of hydrated sodium thiosulphate used we have to remember that water constitutes some of that mass; hence we must use the entire molar mass of the hydrated salt including the “.5H2O”. Hence the Mr value for the calculation is 248.2 (for Na2S2O3.5H2O) and not 158.2 (for Na2S2O3 only). When the solid dissolves and the lattice breaks up, the water molecules in the “.5H2O” go off to become part of the water in the solution, but they are still present when the solid is weighed.
Example mass measurements are shown in the diagram below and the final calculation of the solution concentration are in the box that follows:

The amount of water in solutions
As a final point on solutions, which I never tire of going on about, consider the quantity of water used to prepare these solutions. Pure water has a density of 1.0 g cm-3 at 25OC (i.e. every cm3 of water has a mass of 1.0 g). This means that in Example 1, almost 100g of water would have been used in the since the solution volume was 100 cm3. By contrast only 1.177 g of NaCl was used, just a little over 1% of the total mass of the solution. A similar situation was described in Example 2; where 2.479 g sodium thiosulphate was used compared to around 250 g of water. And these amounts will be fairly typical of many of the solutions used in A-level chemistry. Hence, solutions are nearly all water and this is vital for understanding many practical situations in chemistry.
Copyright Simon Colebrooke 22nd January 2026
Suggested next pages:
 chemistryexplained.uk
chemistryexplained.uk

