Previous sections have described how some aspects of covalent bonding can be understood by thinking about the electrostatic forces between charged sub-atomic particles in the bonding atoms (electrons and protons in nuclei). They have also introduced the “octet rule” which is often useful for helping us decide how many covalent bonds different atoms will form, although this doesn’t always work.
In order to go on and study properties of covalent substances such as the angle between bonds, the shapes of molecules, reactivity and colour, it is necessary to further develop an understanding of covalent bonding. The best way to do that is by thinking about which orbitals from each atom are going to be involved with the bond and how they will interact with one another. This will also help to rationalise things like the “octet rule” and avoid us having to “just accept” that molecules tend to follow it.
Atomic orbitals
Remember that the majority of the volume within an atom consists of the electron orbitals, which are regions of space in which there is a high probability of finding electrons. Alternatively, orbitals are “electron clouds” filling up specific regions of the space. Because they are associated with only a single atom, these orbitals are usually called “atomic orbitals”. Orbitals have a definite, set energy and a specific shape; for example s-orbitals have spherical symmetry whereas p-orbitals are more like a figure of eight shape, with the nucleus located between two regions of electron cloud. Orbitals can be empty (no electrons) or can contain one electron or the maximum of 2 electrons, provided that the electrons have opposite spins.
Orbitals and covalent bonds
Thinking about atomic orbitals can help us understand some new aspects of covalent bonding:
1. Covalent bonds form when atomic orbitals overlap
This is a first useful insight – when two atoms approach each other and the distance between them gets smaller, atomic orbitals from the individual atoms will begin to merge / overlap if part of the orbital volume lies between the two atoms. This is shown in the diagram below, where the region inside the blue ellipse is common to both the atomic orbitals on atoms 1 and 2:
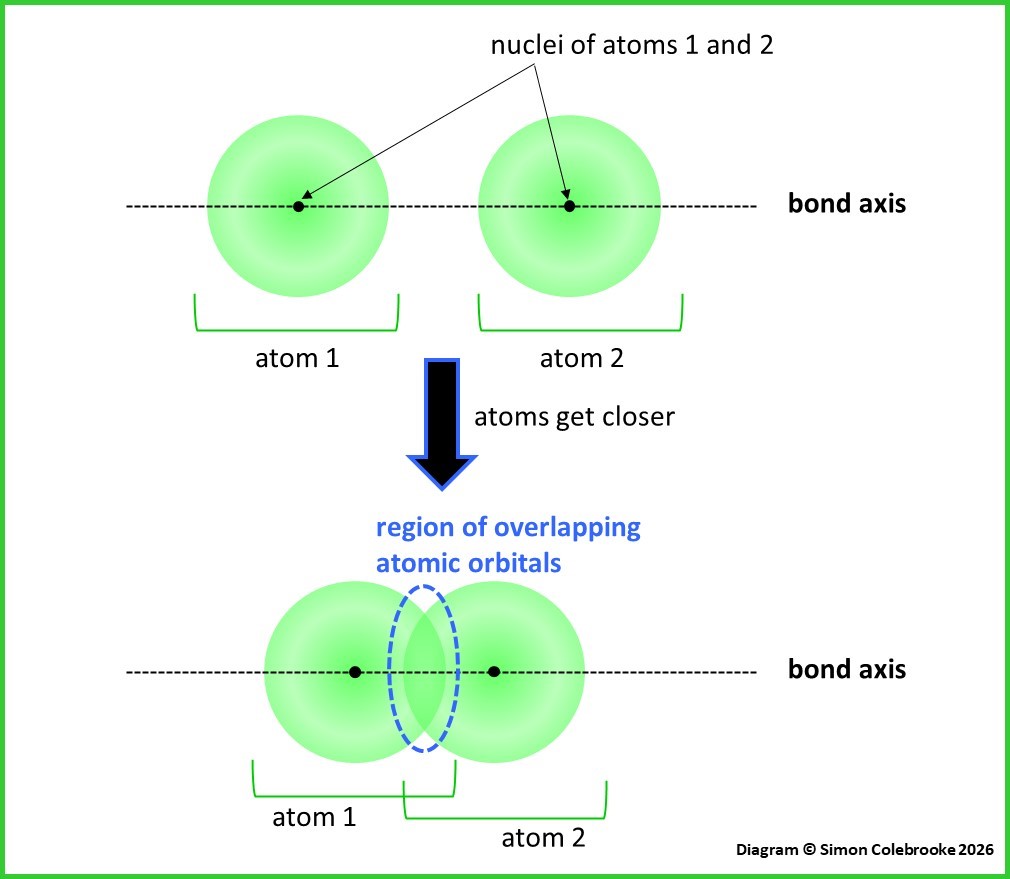
We can imagine the orbitals overlapping along an imaginary line or “axis” that connects the nuclei of the two atoms. This means that the covalent bond will have a specific location / direction within the molecule as the overlap will be between the two bonding atoms. It will be worth contrasting this with ionic and metallic bonds, described later, which are not so directional and thus make covalent bonds unique.
2. The overlapping atomic orbitals usually contain 1 electron each
Covalent bonds involve shared pairs of electrons and the vast majority of covalent bonds encountered at A-level will be formed from bonded atoms each contributing one electron to the shared pair. This was represented in the dot-cross diagrams as one electron of each symbol type (i.e. one dot and one cross) in the bond. One way of understanding the bond is therefore to imagine the shared pair of electrons arising from the overlap of two atomic orbitals containing one electron each, with one orbital coming from each of the bonded atoms. This is shown below:
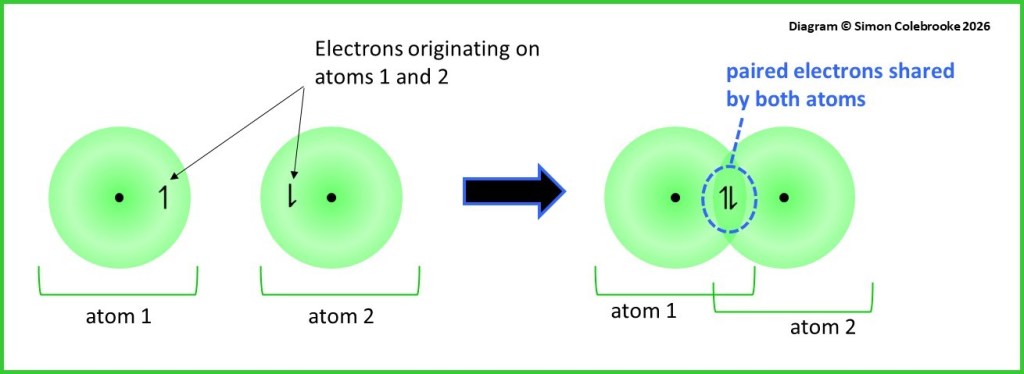
We can now imagine the over-lapped region containing 2 electrons, which is the bonded pair.
This insight will help us identify which atomic orbitals (and maybe also how many) are going to be involved in the formation of covalent bonds; usually those that contain a single, unpaired electron. Hence if we consider a hydrogen atom, with the electron configuration 1s1 it will be the spherically shaped 1s orbital which is involved in the formation of a covalent bond between the hydrogen and another atom. In addition, there is only one of these orbitals per hydrogen atom and hence only one covalent bond can form per hydrogen atom.
This idea will be further developed by “hybridisation” of atomic orbitals to account for the number of bonds different elements form. It is possible for covalent bonds to form in a situation where one of the bonding atoms supplies both the shared electrons. This is called a “dative” bond. Both these aspects will be described in more detail in later sections.
3. Atomic orbitals can overlap in different ways
Different types (shapes) of atomic orbitals can overlap in different ways with those of a neighbouring atom, in terms of symmetry. This results in the formation of different sorts of covalent bonds. In some cases, which type of bond forms will depend upon the relative orientation of the two atomic orbitals and changing that will change the type of covalent bond. This has significant implications for the behaviour and dynamic behaviour of molecules.
The two most common types of covalent bonds are:
Sigma bonds
Sigma bonds (given the symbol σ) are the type of covalent bond that arise when two atomic orbitals overlap in a single region between the two atoms. The overlap region in this case is symmetrically spread around the axis of the bond. Two examples are shown in the diagram below, in Example A the overlap of two spherical s-orbitals and in Example B, two p-orbitals which are overlapping “end-on”. Many other combinations can also lead to a sigma bond.
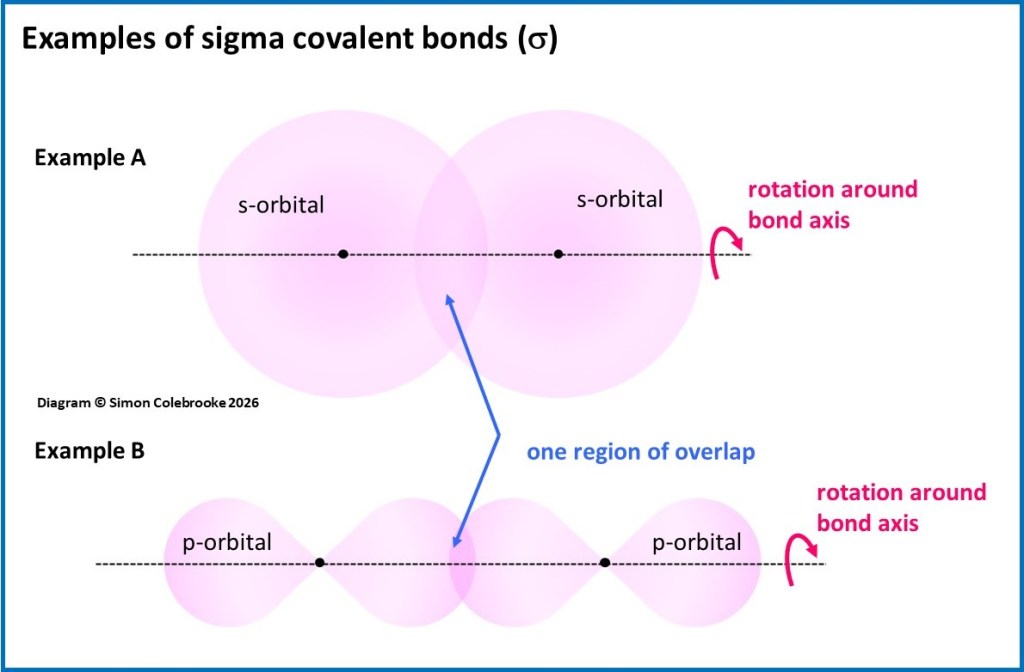
Orbital overlap with this type of symmetry means that it is possible to completely rotate one of the bonded atoms around the bond axis without affecting how the orbitals overlap. Because of the symmetry around the axis, the rotation doesn’t make any difference to how much the orbitals overlap. In the diagram imagine rotating the orbital on the right of each example around the bond axis, as shown by the pink arrow…. nothing changes in the overlap region. Hence, this type of bond can usually be rotated very easily in a real molecule (only a tiny amount of energy is needed to do it) and so parts of molecules connected by sigma bonds are quite flexible. Single covalent bonds are nearly always sigma bonds.
Pi bonds
By contrast, some atomic orbitals have a shape which means they can potentially overlap with an orbital on a neighbouring atom in two distinct regions. The covalent bond formed by this kind of overlap is called a “pi” bond, given the symbol π. An example of two p-orbitals overlapping with a “side on” orientation is shown in the diagram below and results in two overlapping regions, one above and one below the bond axis.
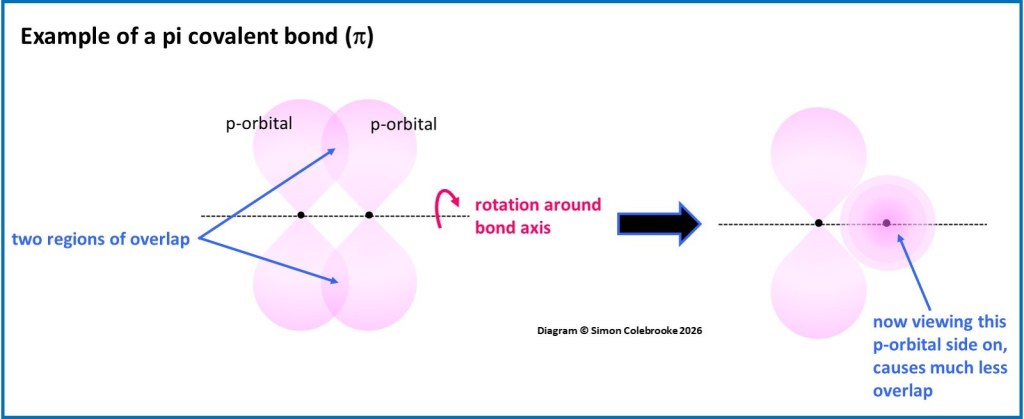
The symmetry of this type of overlap is quite different to that of a sigma bond and means that rotating one of the atoms around the bond axis would completely change the amount of overlap between the two orbitals. For example, rotating the p-orbital on the right by 90O would give almost no overlap (see the diagram). This means that rotation effectively breaks the pi-bond and so usually, the amount of energy required for this to occur is too high for it to happen normally. Hence, pi bonds are less flexible than sigma bonds and generally cannot be rotated. At least one part of a double (and two parts of a triple bond) are pi bonds, so C=C bonds do not usually rotate. This has lots of implications for double bonded molecules, which will be covered later.
The better the atomic orbitals overlap the stronger the covalent bond; this means that π-bonds are usually weaker than a σ-bond between the same two atoms.
3. Molecular orbitals
A third aspect of covalent bondings involves taking orbital overlap a bit further and modelling what happens if the two overlapping orbitals actually “interfere” with each other to form new “molecular” orbitals. Molecular orbitals are effectively combinations of the overlapping atomic orbitals from the individual atoms. Although not always used that often in A-level chemistry this approach is actually the one that gives the best overall description of a covalent bond, and it will be useful for understanding many features of covalent molecules.
When two atomic orbitals from neighbouring atoms overlap, two new orbitals are formed. The new orbitals cover both atoms and so are called “molecular orbitals” rather than atomic orbitals. The exact shape of the molecular orbitals depends upon which atomic orbitals are used and their orientation; details are not needed at A-level. However, an example is shown for the overlap of two hydrogen 1s orbitals when the H2 molecule forms. Both nuclei are inside the same molecular orbital. This should give you an idea of what occurs even when the exact shapes do not need to be known.
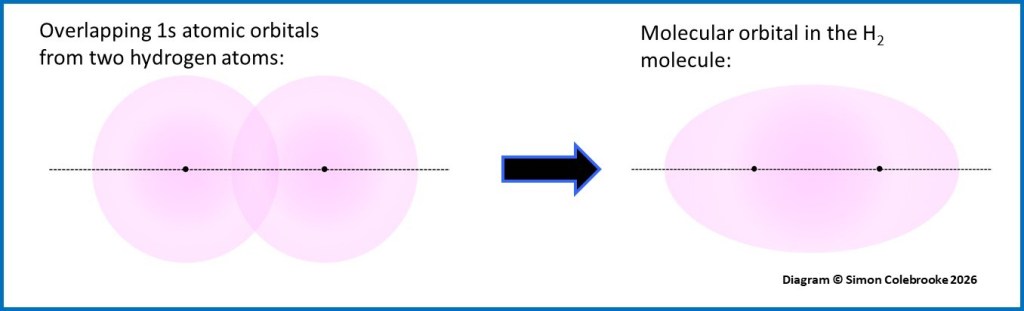
An important point is that when two atomic orbitals overlap, two new molecular orbitals are formed. Of these, one has higher energy than either of the original orbitals and one has lower energy. This is shown in the diagram below for the overlap of two identical 1s orbitals in the formation of the hydrogen molecule:
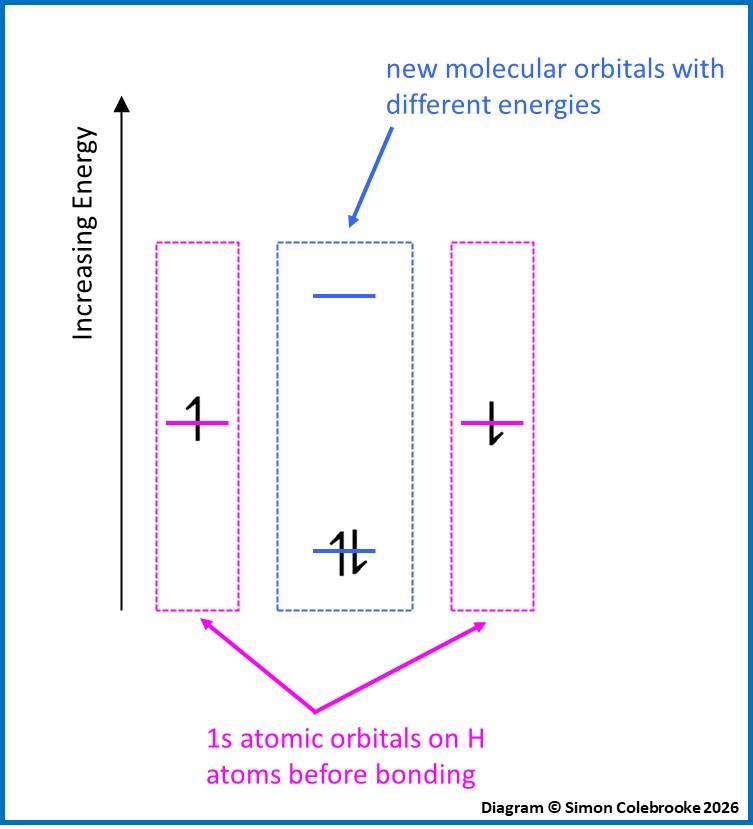
The same rules are used to fill molecular orbitals as was used with atomic orbitals; electrons occupy lowest energy orbitals first and must have paired spins if they are in the same orbital. This means that if each atomic orbital has one electron to begin with the higher energy molecular orbital usually remains empty. As the diagram shows, the electrons end up paired and in a lower energy orbital than either of the original 1s orbitals. This means that the overall energy of the electron arrangement is lower in the molecule than it was for the unbonded atoms; the molecule is more stable than the unbonded atoms. This decrease of energy is why bond forming always releases energy.
The molecular orbital contains electrons from both atoms. Compare that to electrostatic picture of a covalent bond and we can see it is in many ways consistent; the electrons now occupy a region of space containing both nuclei and we can picture how they will be attracted to both at once.
Concern about electron spins
Students often worry about electron spins when covalent bonds form and I can see why. The concern is usually something like this: “what if the electrons in the original atoms are both up-spins? They can’t go into the same orbital because to do that they must have opposite spins”. This is partly correct, because the Pauli Principle states that electrons in the same orbital must have opposite spins. However, because electrons are quantum particles they are not fixed as being either one spin or another and, under most circumstances, there is no difference in energy for a given electron being either spin-up or spin-down. Hence, electrons can flip spins to be paired up in the same molecular orbital and that situation is the only one which gives a low-enough energy to actually occur.
“Copyright Simon Colebrooke 20th February 2026″
Update History
 chemistryexplained.uk
chemistryexplained.uk

