Almost every experiment you will carry out in A-level chemistry will involve using some glassware; whether that is for recording precise measurements, or simply for containing solutions / liquids during a procedure. Chemists use a vast range of glassware for different purposes and there are too many to discuss them all in one go! However, some types of glassware are used extremely frequently, and so thinking about what these are designed for is a good starting point. As you get familiar with different pieces of glassware you will find it easier to decide when it is appropriate to use each type and develop your ability to design experiments.
I find the following categorisation quite useful:
1. Qualitative
This sort of glassware can really only be used as a container. Equipment in this category may not have any sort of scale markings at all, or it might have a very approximate scale – but not one that you could use to make measurements with any degree of certainty. Types of equipment in this group include those described below and drawn schematically in this diagram:
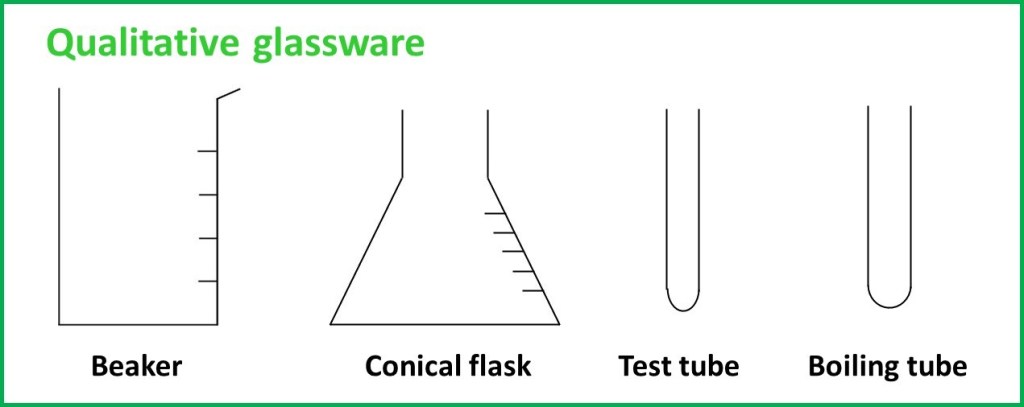
Test tubes – these are thin glass tubes, curved at one end and open at the other. There is no scale printed on them. They are essentially used to hold small samples of liquids or solutions and allow you to watch what happens when reactions are carried out inside on a small scale. They are also often used for collecting small quantities of gas. The test tube can be sealed with a rubber stopper / bung to prevent loss of sample (but never heat a test tube with a bung in!).
Boiling tubes – are much the same as test tubes, but usually much larger diameters (hence larger volumes). As the name implies, they are suited for heating directly (e.g. in a flame) and so are often made from more heat resistant types of glass than test tubes.
Beakers – very open containers, available in a range of sizes, typically from about 25 cm3 to 500 cm3, though larger and smaller options exist. They are useful for a range of functions like mixing reactants, collecting waste or There is often a printed scale on the beaker, but the divisions are very large (maybe 50 cm3) and many also have a disclaimer like “scale is approximate only” printed on the side. The beaker scale is not suitable for anything beyond a ball-park figure, where a 10-20% uncertainty is not an issue. An example might be if you want to use a beaker as a water bath and keep around 100 cm3 of water inside; the scale is probably sufficient for that, but otherwise, beakers are just containers.
Conical flasks – are somewhat like beakers but with slanting sides giving a narrow neck (they are cone shaped). The scale on a conical flask is similarly approximate to a beaker and so, although they are are used in titrations, all the measurements are made by other items of glassware and the flask is just there to hold the solutions. The narrowing neck is ideal for a titration however, because you can swirl and mix the contents without splashing drops out of the sides. Conical flasks are usually referred to as Erlenmeyer flasks in the USA, which is a pretty neat term in my opinion.
Both beakers and conical flasks have flat bases, so they can be used to stand on benches, hot-plates, stirrers and gauzes over a Bunsen burner etc, increasing their versatility. Since the scale is approximate, they are not adversely affected by heating.
2. Graduated scales
Glassware in this category have useful scales printed on them by the manufacturer. They usually have an indication of the quality of the scale as well, but it is always substantially more precise than beakers and conical flasks; you can make useful measurements with these. They usually allow reasonable precision, to nearest cm3 or much less, dependant upon the size of the item.
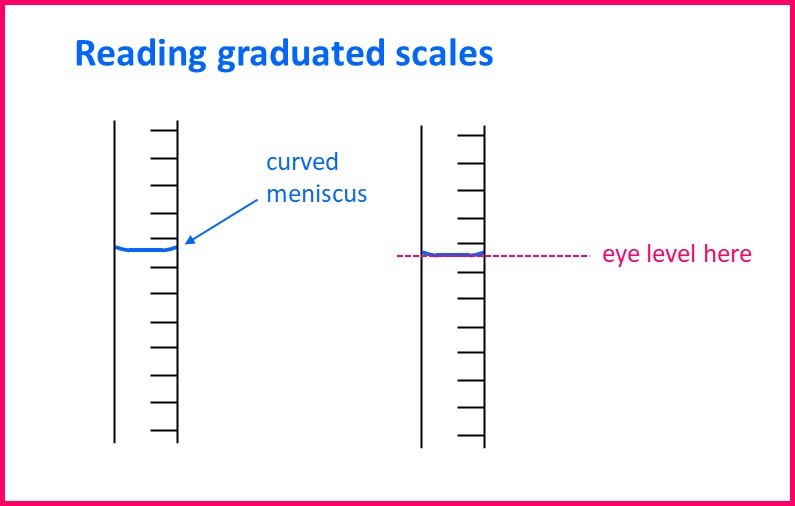
Normally this glassware has quite a small diameter – or has a reasonably narrow “neck” – where the scale is read. It is usually narrow enough that when you read the scale you will be able to see the curved surface of the water / aqueous solution, caused by surface tension. This has the weird term “meniscus”. You always have to read the scale with your eye-level to surface of liquid / solution, and line up the bottom of the meniscus with the scale. More of this in the section on uncertainty, but here is a quick illustration:
Types of equipment in this group are:
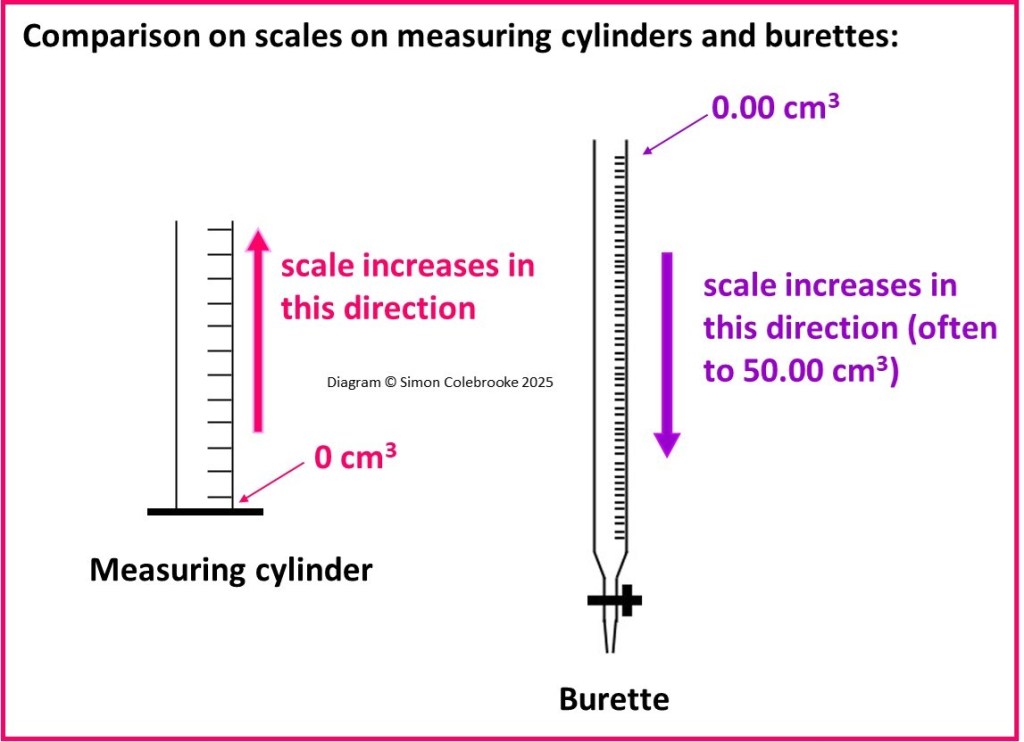
Measuring cylinders – these have a flat base and will stand up-right. They are available in a wide range of sizes those allowing measurement of total volumes of around 10 cm3 up to around 1000 cm3 are quite common. Their major use is measuring out quantities of liquids or solutions to use in a reaction, and so there is usually a spout making it very easy to pour a liquid / solution from a measuring cylinder into another container.
Always choose the measuring cylinder which has the closest total volume to the quantity you want to measure – this keeps the uncertainty as low as possible. For example, if you need 6 cm3 of solution use a 10cm3 measuring cylinder rather than a 50cm3 cylinder, even if the latter has a scale on which you can measure 6 cm3.
Burettes – are rather like a measuring cylinder in someways, but they need to be clamped upright. There is a tap at the bottom, which can be turned to allow solution to run through the burette and the rate of this can be carefully controlled; solutions can be added to another container one drop at a time.
To measure the volume of solution delivered by a burette you need to read the scale on two occasions; once near the top to take an initial reading and then again near the bottom of the scale when you finished adding the solution. The difference is the “titre”. Burette scales are more precise than most measuring cylinders, having a total uncertainty per volume measurement (titre) of only 0.10 cm3.
It is also important to notice that the scale is arranged in the opposite direction to a measuring cylinder, with the “0.00 cm3” line on a burette being near the top. Hence, be very careful when reading scales to check which direction indicates increasing volume. This is indicated in the diagram below:
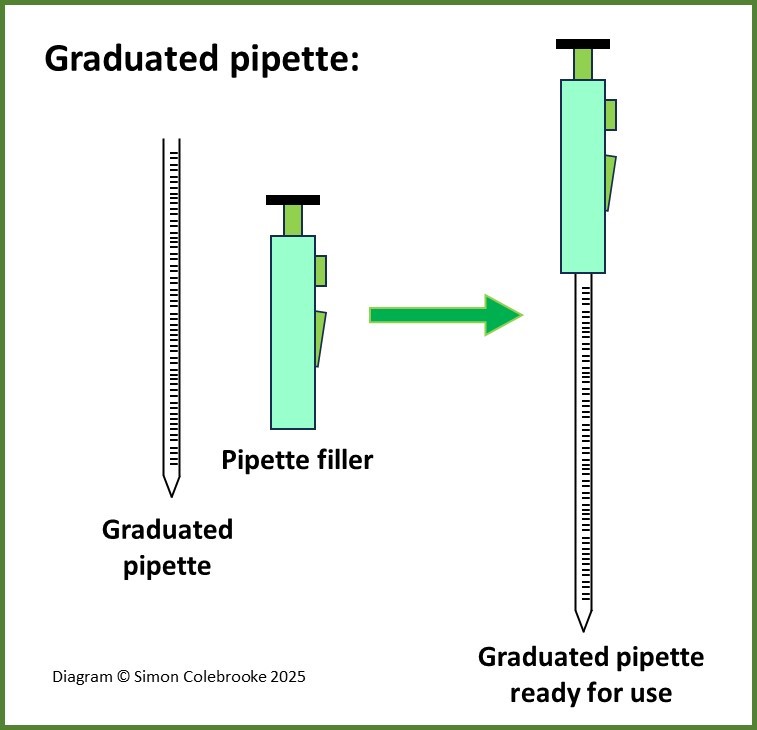
Graduated pipettes – are thin glass tubes with a fine tip, into which you can draw up a solution, reading the graduated scale to determine the volume. Normally a pipette filler of some kind is needed to reduce the air pressure inside the pipette so that the solution is drawn upwards. The filler is attached to the top of the pipette. The pipette is then allowed to drain out its’ contents into another vessel. Graduated pipettes are available to measure a range of different volumes, usually with higher precision than measuring cylinders.
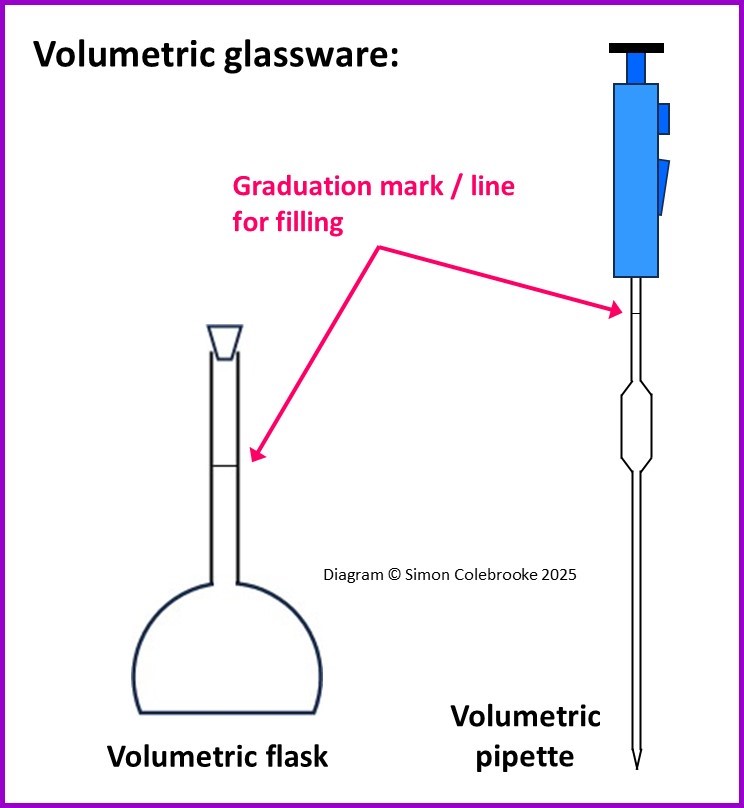
3. Volumetric
This type of glassware has very high precision. There is no graduated scale with multiple divisions, instead there is just a single line etched onto the glass meaning that it can only be used to measure one fixed volume. The solution should fill the container until the bottom of the curved meniscus is aligned with that graduation mark.
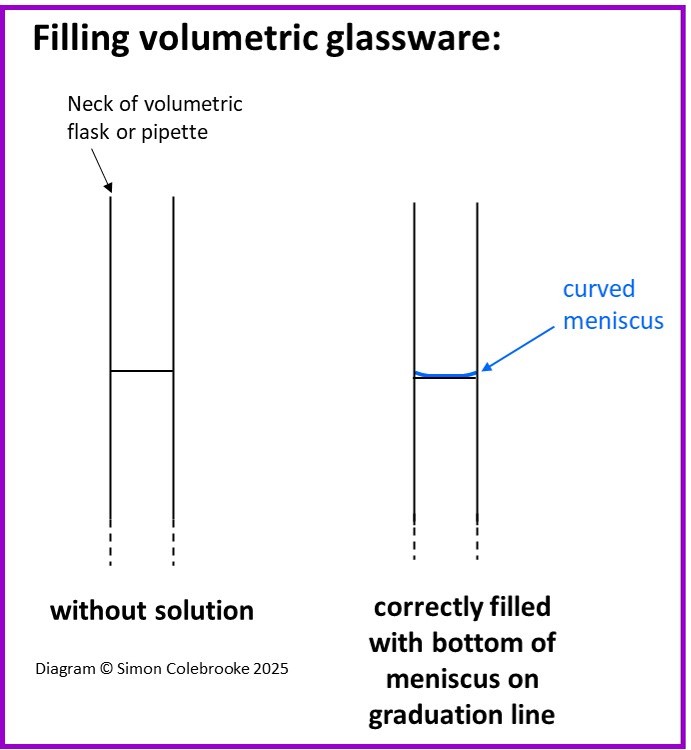
Volumetric glassware is calibrated to contain that one fixed volume at a set temperature. The uncertainty in the measurement is printed on the glassware. Consequently, they cannot be heated as that will alter the shape and volume they contain.
There are two common examples:
Volumetric flasks – these have a very narrow neck where the graduation mark is located and a curved section containing the bulk of the volume. The base is flat however, so that stand easily on benches. Plastic stoppers usually fit into the ground glass top of the flask. These flasks are used to prepare standard solutions of known concentration, but since the flask cannot be heated, the solution is initially prepared in a beaker (where it can be warmed if required) and then transferred to the flask. Volumetric flasks come in a wide range of sizes, from around 5.0 cm3 (or even lesson) to as much as 5000 cm3 for preparing bulk quantities.
Volumetric pipettes – similar to graduated pipettes in usage, but only one graduation line for filling. Volumetric pipettes can only deliver a fixed volume of solution. The most common volumes are 10.0 cm3 and 25.0 cm3, but a huge range exist,, though the available problems are usually multiples of 5.0 or 10.0 cm3. The pipette has a very narrow stem and a bulb that holds the majority of the solution. Pipette fillers are needed to draw solution into the pipette.
4. Quickfit
Items of Quickfit glassware have ground glass joints (opaque smoothed glass) that allow different pieces to be connected. An amazing variety of different components exist including flasks of different shapes, condensers, funnels, thermometer holders and so on. Adapters allow all these different parts to be connected in different arrangements so that numerous complex procedures can be carried out. Typically, Quickfit is used for synthetic reactions to prepare different compounds.
Generally Quickfit items do not have scales on them, so that quantities must be measured using other pieces of equipment. The flasks can be heated, allowing reactions to be performed at higher temperatures.
Next…
This is just a short summary of the range of glassware available in chemistry labs, so that you’ll have a general idea of their role in the sections that follow. More detail on many of these pieces of equipment and how to use them correctly will be described in subsequent sections. Next, you should read about how to make measurements and understand the uncertainty associated with different pieces of glassware and then see how this is applied to common practical procedures.
“Copyright Simon Colebrooke 18th October 2025.”
 chemistryexplained.uk
chemistryexplained.uk

